Emerging Organic Contaminants
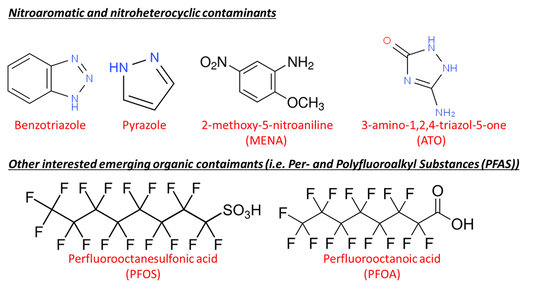
Per- and Polyfluoroalkyl substances (PFAS)
PFAS are a family of anthropogenic chemicals that contain one or more perfluoroalkyl moiety (-CnF2n-) and have been widely used in industrial processes and consumer products due to the thermal and chemical stability as well as the hydro- and oleophobicity of the perfluoroalkyl moiety. The unique physiochemical properties of PFAS also make them highly mobile and persistent in the environment with high bioaccumulation potential. The presence of PFAS in all types of waters and human bodies through the world has been documented in literature. As concerns arise for their adverse effects, including hepatic, developmental, immune, neurobehavioral, endocrine, and metabolic toxicity in human, especially in infants and children, combined efforts have been made by federal and state regulatory agencies to eliminate the manufacturing and application of PFAS as well as to control the release of PFAS to the environment, especially near drinking water sources. Though, since the early 2000s, the U.S. has set regulations and a control roadmap, and perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) have been phased out by major manufacturers, environmental contamination by and human exposure to PFAS are expected to continue due to their high persistence and bioaccumulation potential, and the fact that other PFAS, especially shorter chain PFAS (C4-6), are still in use and will not be replaced in the short term. In 2016, the U.S. EPA set a health advisory level (HAL) of 70 ng/L for individual/combined PFOA and PFOS in drinking water, while some states (i.e., California and New Jersey) have set even lower notification levels (NL) and maximum contaminant levels (MCL). New laws with stricter limits for individual or group PFAS are under discussion and expected to be implemented in the future. Therefore, development of efficient treatment techniques for removing PFAS is highlighted in both the engineering and public health fields to secure water quality and environmental safety for our society.
Nitroaromatic and Nitroheterocyclic Contaminants
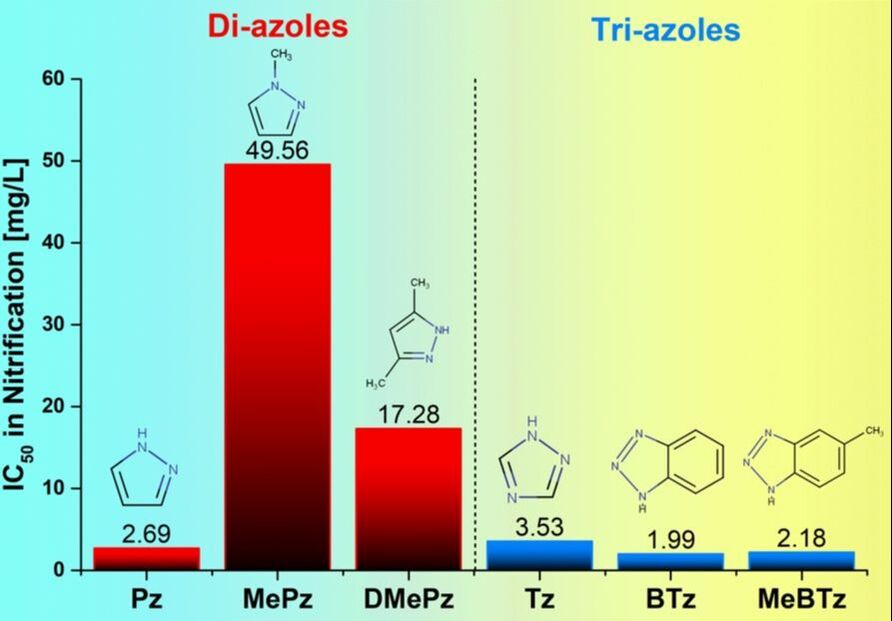
Diazoles and triazoles are N-heterocyclic compounds containing a five-member ring with either 2 or 3 nitrogen atoms, respectively. These azoles and their derivatives are typical emerging contaminants (ECs) used in a wide variety of applications ranging from pesticides and pharmaceuticals to plastics to anticorrosion agents applied to aircraft deicing formulations, cooling tower water and dishwashing tablets. Benzotriazole and its derivatives are used in large quantities as corrosion inhibitors in water treatment systems and semiconductor manufacturing, as well as for UV-light stabilizer for plastics, and antifoggant in photography. They are found among the highest levels of ECs in the environment and waste effluents (up to mg L−1 levels in extreme cases). 1,2,4-Benzotriazole, for instance, is listed as a High Production Volume (HPV) chemical by the U.S. Environmental Protection Agency (EPA).
Collaborators:
Nutrient Removal and Recovery
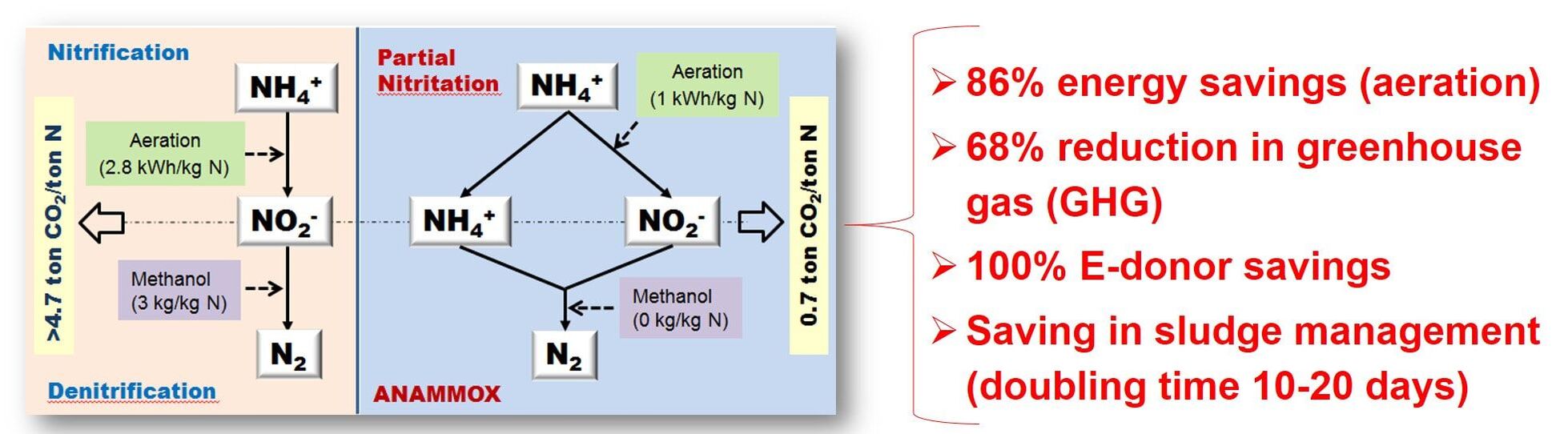
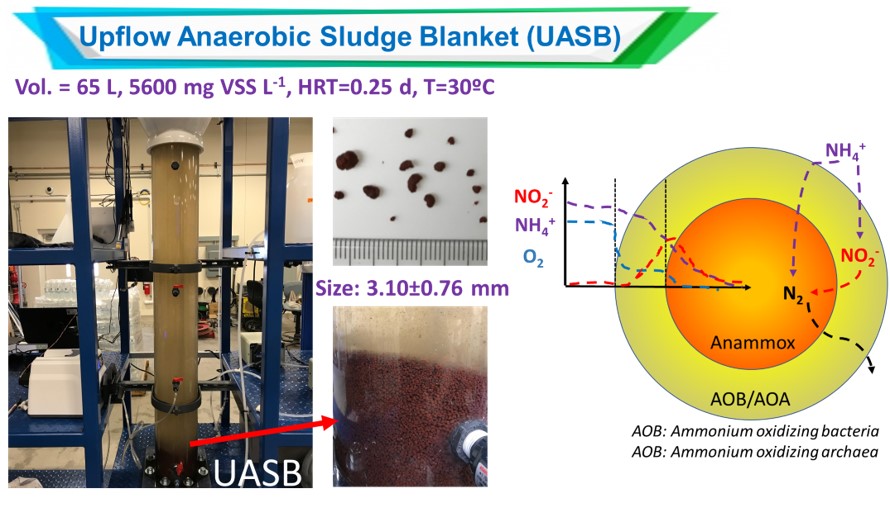
Anaerobic ammonium oxidizing bacteria (Anammox) are known to utilize ammonium and nitrite as electron donor and accepter, respectively, to produce nitrogen gas as their main final product with by-product formation of nitrate. Anammox bacteria provide the advantages of significant saving in aeration, no requirement for external electron donor, reduction of greenhouse gas emission, lowered sludge production, and higher specific nitrogen-removing activity compared to the conventional nitrification-denitrification process used in nutritent-N removal. Therefore, anammox process has recently been widely studied and applied as a state-of-the-art biotechnology to remove nutrient nitrogen from ammonium-rich wastewater.
Nitrogen
Although the existence of biological ammonium (NH4+) oxidation using nitrate (NO3-) or nitrite (NO2-) as the electron acceptor under anaerobic condition was believed to be feasible from the evolutionary and thermodynamic perspective in 1970s, the first evidence to support this prediction was reported until 1995, confirming a biological process, patented with the name “anaerobic ammonium oxidation (Anammox)”, was responsible for the NH4+ disappearing coupled with NO2- consumption and dinitrogen gas (N2, major) and NO3- (minor) production. Anamamox bacteria are clustered and identified as Planctomycete, and up to now, include seven genus and at least 22 species which are all uncultured microorganisms (Candidatus). Especially, the widespread anammox process has been demonstrated to play a significant role in the global nitrogen cycle. Researchers reported that anammox accounted for 24-67% of the total N2 production at two continental shelf sites, which explained the phenomenon of unexplainable loss of NH4+ from anoxic marine environments.
Unlike other bacteria, some unique characteristics of anammox bacteria have been observed, making them owe special position in the N-cycle.
Unlike other bacteria, some unique characteristics of anammox bacteria have been observed, making them owe special position in the N-cycle.
- A large volume of the anammox bacteria cell is occupied by a membrane-bound intracellular compartment named “anammoxosome”, in which the electron-transport phosphorylation machinery is localized as well as other catabolic reactions of the anammox pathway takes place.
- Hydrazine (N2H4), an extremely reactive and highly toxic compound, is formed from the reduction of NO2- and used as a metabolic fuel to drive the electron transport chain that creates a proton gradient across the anammoxosome membrance driving adenosine 5’-triphosphae (ATP) synthesis. The forging of the N-N bond in N2H4 is catalyzed by hydrazine synthase (HZS), a fairly slow enzyme and its low activity possibly explaining the slow growth rates and long doubling times (11 to 22 days) of the anammox bacteria.
- The membrane of the anammoxosome consist of glycerolipid bilayer of ladder-like (“ladderane”) cyclobutane and cyclohexane ring structures that form a strong barrier and serve to limit the transmembrane diffusion of toxic or valuable metabolites (i.e., N2H4) from anammox catabolism and slow the breakdown of the proton gradient used to synthesize ATP during the slow anammox metabolism.
Collaborators
Phosphorus
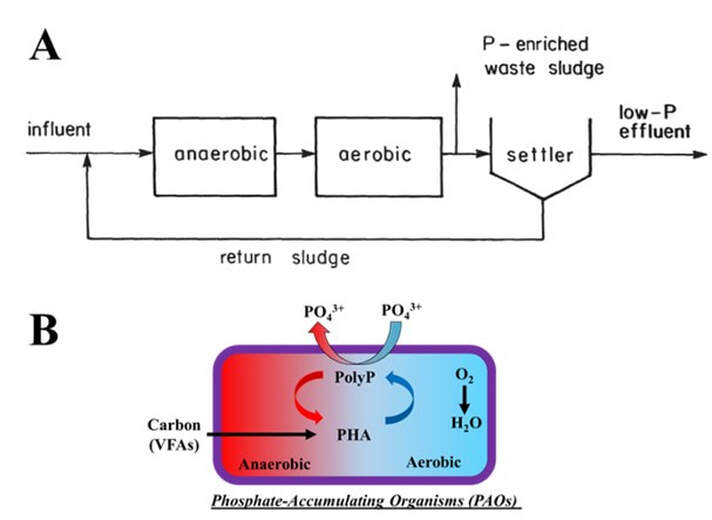
Enhanced biological phosphorus removal (EBPR) has been applied worldwide as an environmentally sustainable and cost-effective technology to achieve low effluent P and biological P removal by cycling phosphate-accumulating organisms (PAOs) capable of P-uptake between carbon-feasting (anaerobic) and carbon-famine (aerobic/anoxic) conditions. A basic EBPR process is shown in Fig. 1A, while the metabolic pathways of PAOs in EBPR process is illustrated in Fig. 1B. Volatile fatty acids (VFAs), as carbon source, are taken up by PAOs anaerobically and stored as polyhydroxyalkanoates (PHAs) using energy generated mostly from hydrolysis of polyphosphate (PolyP) and partly from degradation of glycogen (not shown). In the subsequent aerobic condition, P is taken up to replenish the PolyP reserve accompanied by intracellular organic matter degradation for biomass growth. P removal is achieved via removal of PAO biomass with the waste activated sludge. Although other PAO (i.e., Tetrasphaera, Acinetobacter, etc.) have been reported, Candidatus Accumulibacter phosphatis (Accumulibacter) is the most widely studied PAO and is the basis of most metabolic models. Current results show that incorporation of 90% of the influent P mass into biosolids can be achieved in a stable EBPR process, resulting in a higher P content (2–5% by dry mass) in biosolids. The P-rich biosolids can be used directly as slow release fertilizer via land application in agriculture or to produce other fertilizer products such as struvite (NH4MgPO4·6H2O). This can take place via additional treatments (i.e. P release as orthophosphate (Ortho-P) in an anaerobic digester, followed by chemical precipitation in presence of ammonium and magnesium at the proper pH). Other advantages of applying EBPR include reduction in biosolids weight and volume, reduced plant energy cost for aeration and thereby a reduced carbon footprint.
Collaborators
PUBLICATIONS
1. Rodriguez-Freirea L, Gonzalez-Estrellab J, Li G, Chapter 15: Technologies for fractionation of wastewater and resource recovery, Wastewater Treatment Residues as Resources for Biorefinery Products and Biofuels, Elsevier, 2020, ISBN: 978-0-12-816204-0
2. Li G; Field, J. A.; Zeng, C.; Madeira, C. L.; Nguyen, C. H.; Jog, K. V.; Speed, D.; Sierra-Alvarez, R., Diazole and triazole inhibition of nitrification process in return activated sludge. Chemosphere 2019, 124993.https://doi.org/10.1016/j.chemosphere.2019.124993;[IF=4.068]
3. Lakhey N, Li G, Sierra-Alvarez R, Field JA, Toxicity of azoles towards the anaerobic ammonium oxidation (anammox) process. J Chem Technol Biotechnol. 2019, doi:10.1002/jctb.6285. [IF=2.744]
4. Camila L. Madeira, Warren M. Kadoya, Li G, Stanley Wong, Reyes SierraAlvarez, Jim A. Field. Reductive biotransformation as a pretreatment to enhance In Situ chemical oxidation of nitroaromatic and nitroheterocyclic explosives. Chemosphere. 2019: https://doi.org/10.1016/j.chemosphere.2019.01.178 [IF=4.068]
5. Raju Khatiwada, Leif Abrell,Li G, Robert A Root, Reyes Sierra-Alvarez, James A Field, Jon Chorover. Adsorption and oxidation of 3-nitro-1, 2, 4-triazole-5-one (NTO) and its transformation product (3-amino-1, 2, 4-triazole-5-one, ATO) at ferrihydrite and birnessite surfaces. Environmental Pollution 2018 ;240:200-208.[IF=5.291]
6. Li G, Carvajal-Arroyo JM, Sierra-Ãlvarez R, Field JA. Mechanisms and control of nitrite inhibition of anaerobic ammonium oxidation (ANAMMOX). Water Environ. Res., 2017: 4 (89): 330-336.[IF=0.842]
7. Gonzalez-Estrella Jorge,Li G, Neely Sarah E., Puyol D, Sierra-Alvarez R, Field JA. Elemental copper nanoparticle toxicity to anaerobic ammonium oxidation and the influence of ethylene diamine-tetra acetic acid (EDTA) on the controlling copper toxicity. Chemosphere, 2017: 184: 730-737.[IF=4.068]
8. Li G, Sierra-Alvarez R, Vilcherrez D, Weiss S, Gill C, Krzmarzick MJ, Abrell L, Field JA. Nitrate reverses severe nitrite inhibition of anaerobic ammonium oxidation (anammox) activity in continuously-fed bioreactors. Environ. Sci. Technol., 2016: 50 (19): 10518–10526. [IF=6.396]
9. Li G, Vilcherrez D, Carvajal-Arroyo JM, Sierra-Alvarez R, Field JA. Exogenous nitrate attenuate nitrite toxicity to anaerobic ammonium oxidizing (anammox) bacteria. Chemosphere. 2015: 144:2360-2367. [IF=4.068]
10. Li G, Puyol D, Carvajal-Arroyo JM, Sierra-Alvarez R, Field JA. Inhibition of anaerobic ammonium oxidation by heavy metals. J Chem Technol Biotechnol. 2014: 90(5):830-837. [IF=2.744]
11. Carvajal-Arroyo JM, Puyol D, Li G, Swartwout A, Sierra-Ãlvarez R, Field JA. Starved anammox cells are less resistant to NO2− inhibition. Water Res. 2014 ;65(0):170-176. [IF=6.796]
12. Carvajal-Arroyo JM, Puyol D, Li G, Sierra-Ãlvarez R, Field JA. The intracellular proton gradient enables anaerobic ammonia oxidizing (anammox) bacteria to tolerate NO2− inhibition. J Biotechnol. 2014;192:265-267. [IF=2.446]
13. Carvajal-Arroyo JM, Puyol D, Li G, Lucero-Acuña A, Sierra-Ãlvarez R, Field JA. Pre-exposure to nitrite in the absence of ammonium strongly inhibits anammox. Water Res. 2014 48(0):52-60. [IF=6.796]
14. Carvajal-Arroyo JM, Puyol D, Li G, Sierra-Alvarez R, Field JA. The role of pH on the resistance of resting- and active anammox bacteria to NO2− inhibition. Biotechnol Bioeng. 2014 111(10):1949-56. [IF=4.393]
15. Puyol D, Carvajal-Arroyo JM, Li G, Dougless A, Fuentes-Velasco M, Sierra-Alvarez R, et al. High pH (and not free ammonia) is responsible for Anammox inhibition in mildly alkaline solutions with excess of ammonium. Biotechnol Lett. 2014 36(10):1981-1986. [IF=1.809]
16. Wang A, Liu G, Huang J, Wang L, Li G, Su X, et al. Styrene process condensate treatment with a combination process of UF and NF for reuse. J Hazard Mater. 2013 244–245(0):457-462. [IF=5.641]
2. Li G; Field, J. A.; Zeng, C.; Madeira, C. L.; Nguyen, C. H.; Jog, K. V.; Speed, D.; Sierra-Alvarez, R., Diazole and triazole inhibition of nitrification process in return activated sludge. Chemosphere 2019, 124993.https://doi.org/10.1016/j.chemosphere.2019.124993;[IF=4.068]
3. Lakhey N, Li G, Sierra-Alvarez R, Field JA, Toxicity of azoles towards the anaerobic ammonium oxidation (anammox) process. J Chem Technol Biotechnol. 2019, doi:10.1002/jctb.6285. [IF=2.744]
4. Camila L. Madeira, Warren M. Kadoya, Li G, Stanley Wong, Reyes SierraAlvarez, Jim A. Field. Reductive biotransformation as a pretreatment to enhance In Situ chemical oxidation of nitroaromatic and nitroheterocyclic explosives. Chemosphere. 2019: https://doi.org/10.1016/j.chemosphere.2019.01.178 [IF=4.068]
5. Raju Khatiwada, Leif Abrell,Li G, Robert A Root, Reyes Sierra-Alvarez, James A Field, Jon Chorover. Adsorption and oxidation of 3-nitro-1, 2, 4-triazole-5-one (NTO) and its transformation product (3-amino-1, 2, 4-triazole-5-one, ATO) at ferrihydrite and birnessite surfaces. Environmental Pollution 2018 ;240:200-208.[IF=5.291]
6. Li G, Carvajal-Arroyo JM, Sierra-Ãlvarez R, Field JA. Mechanisms and control of nitrite inhibition of anaerobic ammonium oxidation (ANAMMOX). Water Environ. Res., 2017: 4 (89): 330-336.[IF=0.842]
7. Gonzalez-Estrella Jorge,Li G, Neely Sarah E., Puyol D, Sierra-Alvarez R, Field JA. Elemental copper nanoparticle toxicity to anaerobic ammonium oxidation and the influence of ethylene diamine-tetra acetic acid (EDTA) on the controlling copper toxicity. Chemosphere, 2017: 184: 730-737.[IF=4.068]
8. Li G, Sierra-Alvarez R, Vilcherrez D, Weiss S, Gill C, Krzmarzick MJ, Abrell L, Field JA. Nitrate reverses severe nitrite inhibition of anaerobic ammonium oxidation (anammox) activity in continuously-fed bioreactors. Environ. Sci. Technol., 2016: 50 (19): 10518–10526. [IF=6.396]
9. Li G, Vilcherrez D, Carvajal-Arroyo JM, Sierra-Alvarez R, Field JA. Exogenous nitrate attenuate nitrite toxicity to anaerobic ammonium oxidizing (anammox) bacteria. Chemosphere. 2015: 144:2360-2367. [IF=4.068]
10. Li G, Puyol D, Carvajal-Arroyo JM, Sierra-Alvarez R, Field JA. Inhibition of anaerobic ammonium oxidation by heavy metals. J Chem Technol Biotechnol. 2014: 90(5):830-837. [IF=2.744]
11. Carvajal-Arroyo JM, Puyol D, Li G, Swartwout A, Sierra-Ãlvarez R, Field JA. Starved anammox cells are less resistant to NO2− inhibition. Water Res. 2014 ;65(0):170-176. [IF=6.796]
12. Carvajal-Arroyo JM, Puyol D, Li G, Sierra-Ãlvarez R, Field JA. The intracellular proton gradient enables anaerobic ammonia oxidizing (anammox) bacteria to tolerate NO2− inhibition. J Biotechnol. 2014;192:265-267. [IF=2.446]
13. Carvajal-Arroyo JM, Puyol D, Li G, Lucero-Acuña A, Sierra-Ãlvarez R, Field JA. Pre-exposure to nitrite in the absence of ammonium strongly inhibits anammox. Water Res. 2014 48(0):52-60. [IF=6.796]
14. Carvajal-Arroyo JM, Puyol D, Li G, Sierra-Alvarez R, Field JA. The role of pH on the resistance of resting- and active anammox bacteria to NO2− inhibition. Biotechnol Bioeng. 2014 111(10):1949-56. [IF=4.393]
15. Puyol D, Carvajal-Arroyo JM, Li G, Dougless A, Fuentes-Velasco M, Sierra-Alvarez R, et al. High pH (and not free ammonia) is responsible for Anammox inhibition in mildly alkaline solutions with excess of ammonium. Biotechnol Lett. 2014 36(10):1981-1986. [IF=1.809]
16. Wang A, Liu G, Huang J, Wang L, Li G, Su X, et al. Styrene process condensate treatment with a combination process of UF and NF for reuse. J Hazard Mater. 2013 244–245(0):457-462. [IF=5.641]